A-77636
The A-77636 topic is one of the most relevant and important today. Its implications cover numerous fields and its impact can be felt in different aspects of our lives. From A-77636, through A-77636, to A-77636, this topic arouses interest and controversy in equal measure. In this article, we will analyze in detail the different facets of A-77636 and its influence on today's society. From its origins to its evolution in the present, we will explore every relevant aspect of A-77636 to understand its importance and possible ramifications in the future.
| |
| Names | |
|---|---|
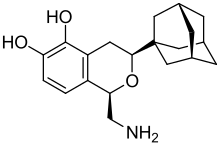
| Preferred IUPAC name
(1R,3S)-3-(Adamantan-1-yl)-1-(aminomethyl)-1H-2-benzopyran-5,6-diol | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C20H27NO3 | |
| Molar mass | 329.440 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C , 100 kPa).
| |
A-77636 is a synthetic drug which acts as a selective D1 receptor full agonist.[1] It has nootropic, anorectic, rewarding and antiparkinsonian effects in animal studies,[2][3][4][5][6] but its high potency and long duration of action causes D1 receptor downregulation and tachyphylaxis,[7][8][9] and unlike other D1 full agonists such as SKF-82,958, it does not produce place preference in animals.[10] A-77636 partially substituted for cocaine in animal studies, and has been suggested for use as a possible substitute drug in treating addiction,[11] but it is better known for its use in studying the role of D1 receptors in the brain.[12][13][14]
References
- ^ Kebabian JW, Britton DR, DeNinno MP, Perner R, Smith L, Jenner P, Schoenleber R, Williams M (December 1992). "A-77636: a potent and selective dopamine D1 receptor agonist with antiparkinsonian activity in marmosets". European Journal of Pharmacology. 229 (2–3): 203–9. doi:10.1016/0014-2999(92)90556-J. PMID 1362704.
- ^ Asin KE, Nikkel AL, Wirtshafter D (February 1994). "Repeated D1 receptor agonist treatment blocks cocaine-induced locomotor activity and c-fos expression". Brain Research. 637 (1–2): 342–4. doi:10.1016/0006-8993(94)91258-0. PMID 7910104. S2CID 20197207.
- ^ Ranaldi R, Beninger RJ (July 1994). "The effects of systemic and intracerebral injections of D1 and D2 agonists on brain stimulation reward". Brain Research. 651 (1–2): 283–92. doi:10.1016/0006-8993(94)90708-0. PMID 7922577. S2CID 18241791.
- ^ Grondin R, Bédard PJ, Britton DR, Shiosaki K (August 1997). "Potential therapeutic use of the selective dopamine D1 receptor agonist, A-86929: an acute study in parkinsonian levodopa-primed monkeys". Neurology. 49 (2): 421–6. doi:10.1212/wnl.49.2.421. PMID 9270571. S2CID 541898.
- ^ Cai JX, Arnsten AF (October 1997). "Dose-dependent effects of the dopamine D1 receptor agonists A77636 or SKF81297 on spatial working memory in aged monkeys". The Journal of Pharmacology and Experimental Therapeutics. 283 (1): 183–9. PMID 9336323.
- ^ Cooper SJ, Al-Naser HA, Clifton PG (February 2006). "The anorectic effect of the selective dopamine D1-receptor agonist A-77636 determined by meal pattern analysis in free-feeding rats". European Journal of Pharmacology. 532 (3): 253–7. doi:10.1016/j.ejphar.2005.11.046. PMID 16478622.
- ^ Lin CW, Bianchi BR, Miller TR, Stashko MA, Wang SS, Curzon P, Bednarz L, Asin KE, Britton DR (March 1996). "Persistent activation of the dopamine D1 receptor contributes to prolonged receptor desensitization: studies with A-77636". The Journal of Pharmacology and Experimental Therapeutics. 276 (3): 1022–9. PMID 8786531.
- ^ Saklayen SS, Mabrouk OS, Pehek EA (October 2004). "Negative feedback regulation of nigrostriatal dopamine release: mediation by striatal D1 receptors". The Journal of Pharmacology and Experimental Therapeutics. 311 (1): 342–8. doi:10.1124/jpet.104.067991. PMID 15175419. S2CID 36903868.
- ^ Ryman-Rasmussen JP, Griffith A, Oloff S, Vaidehi N, Brown JT, Goddard WA, Mailman RB (February 2007). "Functional selectivity of dopamine D1 receptor agonists in regulating the fate of internalized receptors". Neuropharmacology. 52 (2): 562–75. doi:10.1016/j.neuropharm.2006.08.028. PMC 1855220. PMID 17067639.
- ^ Graham DL, Hoppenot R, Hendryx A, Self DW (April 2007). "Differential ability of D1 and D2 dopamine receptor agonists to induce and modulate expression and reinstatement of cocaine place preference in rats". Psychopharmacology. 191 (3): 719–30. doi:10.1007/s00213-006-0473-5. PMID 16835769. S2CID 21192319.
- ^ Chausmer AL, Katz JL (January 2002). "Comparison of interactions of D1-like agonists, SKF 81297, SKF 82958 and A-77636, with cocaine: locomotor activity and drug discrimination studies in rodents". Psychopharmacology. 159 (2): 145–53. doi:10.1007/s002130100896. PMID 11862342. S2CID 6788631.
- ^ Acquas E, Wilson C, Fibiger HC (April 1997). "Nonstriatal dopamine D1 receptors regulate striatal acetylcholine release in vivo". The Journal of Pharmacology and Experimental Therapeutics. 281 (1): 360–8. PMID 9103518.
- ^ Ruskin DN, Rawji SS, Walters JR (July 1998). "Effects of full D1 dopamine receptor agonists on firing rates in the globus pallidus and substantia nigra pars compacta in vivo: tests for D1 receptor selectivity and comparisons to the partial agonist SKF 38393". The Journal of Pharmacology and Experimental Therapeutics. 286 (1): 272–81. PMID 9655869.
- ^ Bubser M, Fadel JR, Jackson LL, Meador-Woodruff JH, Jing D, Deutch AY (June 2005). "Dopaminergic regulation of orexin neurons" (PDF). The European Journal of Neuroscience. 21 (11): 2993–3001. doi:10.1111/j.1460-9568.2005.04121.x. hdl:2027.42/71513. PMID 15978010. S2CID 11527186.