Sultopride
In today's world, Sultopride has gained unprecedented relevance. Whether in the professional, academic or personal sphere, Sultopride has become a topic of common interest for people of all ages and backgrounds. As society advances, the challenges related to Sultopride become more complex, and the need to understand its implications becomes increasingly pressing. In this article, we will explore different aspects of Sultopride and its impact on various areas of daily life. From its history to its current applications, we will address the many facets of Sultopride and its influence on modern society.
 | |
| Clinical data | |
|---|---|
| Trade names | Barnetil, Barnotil, Topral |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral, IM |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Elimination half-life | 3–5 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.053.293 |
| Chemical and physical data | |
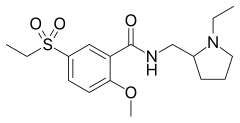
| Formula | C17H26N2O4S |
| Molar mass | 354.47 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Sultopride (trade names Barnetil, Barnotil, Topral) is an atypical antipsychotic of the benzamide chemical class used in Europe, Japan, and Hong Kong for the treatment of schizophrenia.[2][3][4] It was launched by Sanofi-Aventis in 1976.[2] Sultopride acts as a selective D2 and D3 receptor antagonist.[5] It has also been shown to have clinically relevant affinity for the GHB receptor as well, a property it shares in common with amisulpride and sulpiride.[6]
Pharmacology
| Site | Ki | Species | Ref |
|---|---|---|---|
| D2 | 1.6 | Human | [7] |
| D3 | 3.8 | Human | [7] |
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b Miguel Vela J, Buschmann H, Holenz J, Párraga A, Torrens A (2007). Antidepressants, Antipsychotics, Anxiolytics: From Chemistry and Pharmacology to Clinical Application. Weinheim: Wiley-VCH. ISBN 978-3-527-31058-6.
- ^ Swiss Pharmaceutical Society (2000). Index Nominum 2000: International Drug Directory (Book with CD-ROM). Boca Raton: Medpharm Scientific Publishers. ISBN 3-88763-075-0.
- ^ European Drug Index (4th ed.). Boca Raton: CRC Press. 1998. ISBN 3-7692-2114-1.
- ^ Burstein ES, Ma J, Wong S, Gao Y, Pham E, Knapp AE, et al. (December 2005). "Intrinsic efficacy of antipsychotics at human D2, D3, and D4 dopamine receptors: identification of the clozapine metabolite N-desmethylclozapine as a D2/D3 partial agonist". The Journal of Pharmacology and Experimental Therapeutics. 315 (3): 1278–1287. doi:10.1124/jpet.105.092155. PMID 16135699. S2CID 2247093.
- ^ Maitre M, Ratomponirina C, Gobaille S, Hodé Y, Hechler V (April 1994). "Displacement of gamma-hydroxybutyrate binding by benzamide neuroleptics and prochlorperazine but not by other antipsychotics". European Journal of Pharmacology. 256 (2): 211–214. doi:10.1016/0014-2999(94)90248-8. PMID 7914168.
- ^ a b Burstein ES, Ma J, Wong S, Gao Y, Pham E, Knapp AE, et al. (December 2005). "Intrinsic efficacy of antipsychotics at human D2, D3, and D4 dopamine receptors: identification of the clozapine metabolite N-desmethylclozapine as a D2/D3 partial agonist". The Journal of Pharmacology and Experimental Therapeutics. 315 (3): 1278–1287. doi:10.1124/jpet.105.092155. PMID 16135699. S2CID 2247093.